Munitions Constituents - Dissolution
Firing and detonation of munitions in wartime and during live-fire training scatters propellant and explosive particles onto the soil. Once deposited, the energetic compounds in explosives and propellants dissolve in precipitation and interact with soil constituents as they migrate through the subsurface to groundwater. Deposition, dissolution, and transformation of energetic compounds are of interest for two reasons: (1) these chemicals are toxic and can harm humans, animals and plants, and (2) they can migrate through soil and contaminate groundwater. If contaminated groundwater migrates off a military base, political and regulatory actions can close or limit the base to training. Dissolution is thought to be the rate-limiting step for aqueous transport of these compounds to groundwater.
Related Aritcle(s):
CONTRIBUTOR(S): Dr. Susan Taylor
Key Resource(s):
- Dissolution rate, weathering mechanics, and friability of TNT, Comp B, Tritonal, and Octol (No. ERDC/CRREL-TR-10-2)[1]
Introduction
Energetic materials are a class of compound that can store and release chemical energy. High explosives (HE) rapidly release large amounts of gas and energy upon detonation. Insensitive munitions (IM) have similar detonation characteristics as HE, yet do not react to shock and temperature as easily. Propellants do not detonate but burn very quickly, releasing the energy and gas pressure needed to propel bullets and shells to given distances. Because of their properties, these compounds are extensively used by the military and in the construction and mining industry (Table 1)[2].
The explosives used to fill mortar and howitzer shells are generally melt cast, a process where an explosive with a low melting point, such as trinitrotoluene (TNT), is melted, poured into a shell and cooled. To make a multi-component explosive such as Composition B (Comp B), RDX crystals are mixed with molten TNT. For the IM formulations, 2,4-Dinitroanisole (DNAN) is the molten matrix to which crystalline constituents, such as ammonium perchlorate (AP), 3-nitro-1,2,4-triazol-5-one (NTO), and nitroguanidine (NQ) are added.
Explosive Compound Solubility
The mass of energetic compounds that dissolves and enters the soil is a function of many variables such as: 1) the aqueous solubility of the compound, 2) the mass of explosive residues deposited on the soil, 3) the size of individual residues, and 4) the three-dimensional (3D) structure of formulations that have multiple constituents.
The aqueous solubility of a compound determines how quickly it can be dissolved in water. Table 2 lists the solubility for the compounds in various energetic formulations. Note that the nitroaromatics (TNT, 2,4-DNT, and DNAN) and the cyclic nitroamines (RDX and HMX) have low solubility, and the nitrate esters (NG), nitroimines (NQ), nitrotriazole (NTO), and the salt (AP) have progressively higher solubility.
| Energetic | Aqueous Solubility | Formula | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TNT | 100 | C7H5N3O6 | Ro et al., 1996[3] | ||||||||
| RDX | 60 | C3H6N6O6 | Banerjee et al., 1980[4] | ||||||||
| HMX | 4.6 | C4H8N8O8 | Monteil-Rivera et al., 2004[5] | ||||||||
| 2,4-DNT | 280 | C7H6N2O4 | Rosenblatt et al., 1991[6] | ||||||||
| NG | 1500 @ 20°C | C3H5N3O9 | Yinon, 1999[7] | ||||||||
| NQ | 2600±100 | CH4N4O2 | Haag et al., 1990[8] | ||||||||
| DNAN | 276; 213 | C7H6N2O5 | Boddu et al., 2008[9]; Hawari et al., 2015[10] | ||||||||
| NTO | 16642 | C2N4O3 | Spear et al., 1989[11] | ||||||||
| AP | 217000 | NH4ClO4 | Motzer 2001[12] | ||||||||
| Table 2. Aqueous solubility of energetic compounds at 25°C. | |||||||||||
The mass of explosive residues depends on the type of round fired and the manner in which it detonated: high-order, low-order (partial), or not at all, which produces an unexploded ordnance (UXO). High order detonations consume 99.99% of the explosive mass[13] leaving small amounts of µm-size residue[14][15]. In contrast, low-order (LO) detonations deposit some fraction of the explosive fill as particles and chunks in a range of sizes (Fig. 1). UXO contain the entire explosive fill and this is released into the environment when their metal casings are breached. The percentage of fired rounds that undergo these fates varies from one munitions type to another[16]. Dissolution from low-order detonation residue appears to be the largest contributor to present day aqueous transport of explosives from military training[17].
'

The particle sizes of the explosive pieces are important because they control the mass that is dissolved and available for transport into soil. Several factors may influence the size of particles scattered by LO detonations including the mass of explosives in the round, the detonation yield (percent of the explosive that detonated), and the nature of the explosive fill[18].
The three dimensional structure of explosive formulations that contain multiple constituents, such as Comp B and the IM formulations, determines the area in contact with water for each compound. This value, along with the solubility of each compound, controls the dissolution rate. The 3D structure of explosives can be imaged using micro-computed tomography (µCT), an X-ray technique that can discriminate among the different constituents in explosives and map their 3D locations[19].
Propellant Compound Solubility
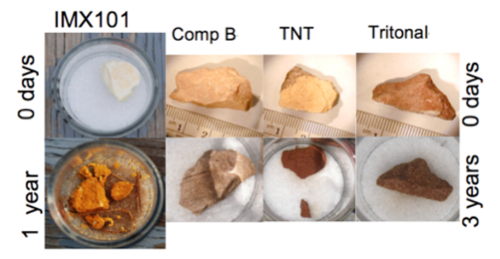
Propellants dissolve differently than do explosives because nitrocellulose (NC), the main constituent of most propellants, is not soluble in water. Consequently the nitrocellulose-based propellants do not change shape as the energetics within them (nitroglycerine (NG), NQ, 2,4-dinitrotoluene (DNT)) leach from their NC matrix[20]. Furthermore, propellants are made to specific sizes and do not break apart. Interestingly, the shape of the original propellant grains dictates the appearance of residues. Un-perforated grains yielded smaller versions of themselves, single perforated grains yielded rings, and multiple perforated grains deposited slivers (Fig. 2).
Measuring Dissolution
Various techniques have been used measure the dissolution of individual HE, IM, and propellant particles both in the laboratory and in outdoor settings. The tests shown in Figure 3 placed pieces of energetic compounds in funnels, collected and analyzed the water for energetics interacting with them and scaled the mass loss to drip rate (lab tests) or precipitation rate (outdoor tests)[21][22][20]. These tests isolated the aqueously dissolved compounds from the confounding effects of soil interaction and have been used to model dissolution of high explosives[23] and insensitive munitions[24]. They more closely simulate the physical circumstances of explosive formulations or propellants exposed to rainfall on surface soils than do stirred-bath[25][26] or glass-bead column experiments[27].

Laboratory Dissolution Tests of HE, IM and Propellants
Dissolution drip tests have been conducted on the high explosives TNT, Comp B, Tritonal, and Octol[28][21], on IM formulations IMX101, IMX104, and PAX21[24], and on a variety of single, double, and triple-based propellants[20] collected after training.
High explosives composed of a single compound such as TNT and Tritonal, retain their original morphologies as they dissolve, and their dissolution time series are fairly simple curves. By comparison, multi-compound explosive particles, such as Comp B and Octol, quickly become lumpy as the surface TNT dissolves, leaving less soluble RDX and HMX crystals on the surface. The Comp B particles become conglomerations of RDX crystals held together by TNT below the surface. Octol has large HMX crystals and only about 30% TNT matrix and these quickly disaggregate into loose HMX crystals when the TNT dissolves (Fig. 4[21]).
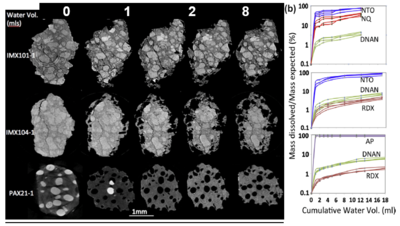
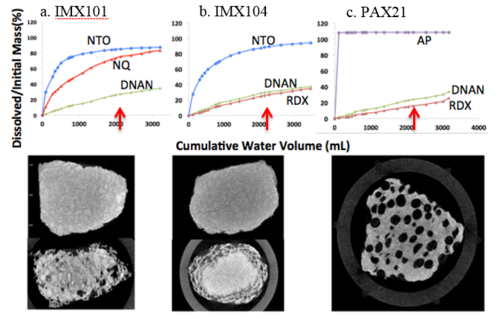
IM formulations dissolve differently. For example, Figure 5a shows three of the 12 IM particles tracked by µCT imaging as they dissolved, and Figure 5b shows the percent mass loss (relative to amount in the formulation) as a function of water volume (four of each formulation). The compounds dissolve in accordance with their solubility. For IMX101, more NTO and NQ dissolve than DNAN, and more NTO dissolve from each particle than NQ despite their being more NQ in the particle. For IMX104, the dissolution order is also in agreement with the solubility of the constituents (Table 2). For PAX21, AP dissolves quickly and completely followed by DNAN and RDX. Solubility is the primary factor that determines the dissolution of these mm-sized IM particles and contact area with water does not appear to limit their dissolution.
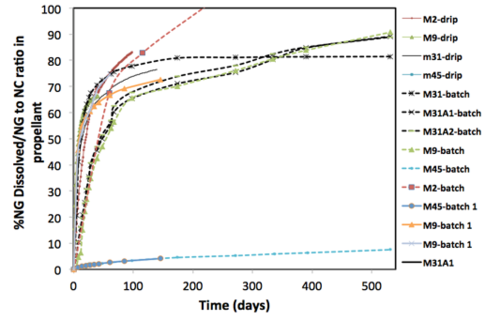
Propellant laboratory tests include both unfired and fired propellant grains. The dissolution versus time curves for 2,4-DNT, NG, and NQ from unfired propellants show a slow, linear loss rate for 2,4-DNT from single-based propellants and much faster dissolution of NG and NQ from double and triple-based propellants. Both the NG and NQ show a fast initial mass loss rate followed by a slower dissolution rate. When the mass of dissolved NG is normalized by nitrocellulose content in the propellant, the mass loss curves for the different propellants cluster. This suggests that the NC binds 10 to 20% of the NG and that any extra NG is lost from the propellants at a similar rate (Fig. 6). An exception from this clustering is the M45 double-base propellant. In this case, the NC may not have been fully nitrated causing it to hold the NG.
Outdoor Dissolution of HE and IM
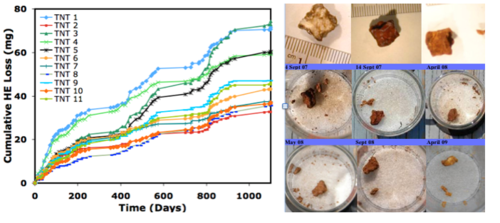
High explosives dissolution has been measured outdoors on pieces of TNT, Comp B, C4, and Tritonal[22][1]. The Comp B, Tritonal, and TNT particles all turned rust red with exposure to sunlight. Four of the 34 HE chunks split naturally during the tests and others spalled small flakes or cracked. An example of the cumulative mass loss curves is shown for the 12 TNT pieces (Fig. 7). Similar data exist for Comp B, Tritonal, and C4[1]. Except for the chunks that split (TNT 3 and 5), the shapes of the cumulative mass loss curves are similar among all the chunks and reflect the seasonal precipitation rate.
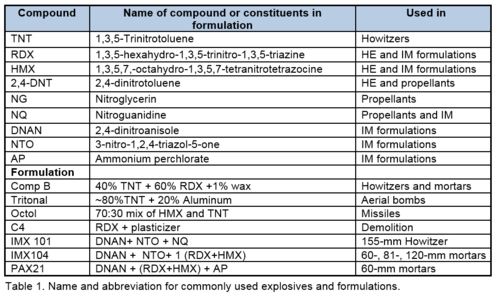
Insensitive Munitions rounds deposit more of their fill onto range soils during high order and blow-in-place detonations than do HE filled rounds[30][31]. IM formulations are also more friable and dissolve more quickly than HE. Figure 8 shows that after just one year an IMX101 piece has crumbled into many pieces whereas pieces of Comp B, TNT and Tritonal are mainly intact after three years of outdoor dissolution and weathering. Their friability might result from fractures seen surrounding the crystal constituents in µCT images of detonated residues[19]. Importantly, this observation also explains why AP, NTO, and NQ are found after ‘high order’ detonations when all of the DNAN is consumed[30][31]: de-bonding of the crystals from the matrix causes them to be scattered instead of detonated. The de-bonded crystals of ammonium perchlorate, a very soluble salt, dissolve quickly and travel in the water. Although not as soluble as AP, the large surface area to volume of the NTO and NQ crystals suggests that these dissolve quickly as well.
As was found in the laboratory tests, IM constituents dissolve in the order of their solubility[19][32]. For example, the averaged mass loss data for five IMX 101 chunks shows that NTO dissolves first, followed by NQ, and finally DNAN (Fig. 9a). For IMX 104, which contains no NQ, NTO is lost first followed by DNAN and RDX (Fig. 9b). Figure 9c shows that the AP in PAX 21 dissolves in the first water sample, leaving DNAN and RDX as the only constituents. The mass loss of AP, NTO and NQ is highest at the start of the test and then decreases, gradually or abruptly (AP), as less of the constituent is available. When the constituent has totally dissolved, it is by definition no longer available and when it is near the center of the particle and shielded from water it is less available. The mass loss of DNAN and RDX, on the other hand, are fairly linear and can be used to estimate the time it will take to completely dissolve particles of these formulations[24].
Summary and Implications for Remediation
Properties of explosive and propellant residues determine how quickly they will dissolve in the environment. High order detonations of explosive-filled shells generally leave very little residue. However, explosives that contain crystalline constituents, such as Comp B and IM formulations, can scatter some fraction of their crystals, even during high-order detonations, if the crystals de-bond from the matrix. Low order detonations of HE and IM-filled munitions can leave large explosive chunks. As HE dissolves slowly, collecting these before they break into small pieces will reduce the amount of dissolved explosives entering the soil. Small pieces both dissolve more quickly and are harder to find. IM formulation, on the other hand, have crystalline components that dissolve very quickly, some have aqueous solubility values that are orders-of-magnitude higher than DNAN, TNT, or RDX. Furthermore, IM formulations are much more friable that HE or propellant residues and more residues are deposited from these rounds when they detonate. Unless the chunks can be collected immediately, some of the soluble constituents such as AP and NTO will enter the soil. Because the formulations are so friable, IM chunks should be collected within a few weeks to avoid having the cm-sized pieces break up into mm-sized pieces. For these reasons, IM formulations are more likely to contaminate groundwater.
Propellant grains are often small and difficult to recover once on the ground. Fortunately, 2,4-DNT, and NQ dissolve fairly slowly as does NG if the propellant has been fired. If the propellant has not been fired, NG can dissolve quickly. Although NG is routinely found in firing range soils, it has not been detected in groundwater below firing ranges. This likely happens because dissolved NG is broken down in soils and the NG retained in the propellant residues needs to diffuse to the surface of the grain to be dissolved - a very slow process. Soils at firing ranges can have high NG concentrations because, during the analyses of soils, NG is extracted from the propellant using organic solvents.
References
- ^ 1.0 1.1 1.2 1.3 1.4 Taylor, S., Lever, J., Walsh, M., Fadden, J., Perron, N., Bigl, S., Spanggord, R., Curnow, M., Packer, B., 2010. Dissolution rate, weathering mechanics, and friability of TNT, Comp B, Tritonal, and Octol (No. ERDC/CRREL-TR-10-2). Engineer Research and Development Center Hanover NH Cold Regions Research And Engineering Lab. ER-1482
- ^ Jenkins, T.F., Hewitt, A.D., Grant, C.L., Thiboutot, S., Ampleman, G., Walsh, M.E., Ranney, T.A., Ramsey, C.A., Palazzo, A.J., Pennington, J.C., 2006. Identity and distribution of residues of energetic compounds at army live-fire training ranges. Chemosphere, 63(8), 1280-1290. doi: 10.1016/j.chemosphere.2005.09.066
- ^ Ro, K.S., Venugopal, A., Adrian, D.D., Constant, D., Qaisi, K., Valsaraj, K.T., Thibodeaux, L.J., Roy, D., 1996. Solubility of 2, 4, 6-trinitrotoluene (TNT) in water. Journal of Chemical & Engineering Data, 41(4), 758-761. doi: 10.1021/je950322w
- ^ Banerjee, S., Yalkowsky, S.H., Valvani, C., 1980. Water solubility and octanol/water partition coefficients of organics. Limitations of the solubility-partition coefficient correlation. Environmental Science & Technology, 14(10), 1227-1229. doi: 10.1021/es60170a013
- ^ Monteil-Rivera, F., Paquet, L., Deschamps, S., Balakrishnan, V.K., Beaulieu, C., Hawari, J., 2004. Physico-chemical measurements of CL-20 for environmental applications: Comparison with RDX and HMX. Journal of Chromatography A, 1025(1), 125-132. doi: 10.1016/j.chroma.2003.08.060
- ^ Rosenblatt, D.H., Burrows, E.P., Mitchell, W.R., Parmer, D.L., 1991. Organic explosives and related compounds. In Anthropogenic Compounds. Springer Berlin Heidelberg. pgs. 195-234. doi:10.1007/978-3-540-46757-1_4
- ^ Yinon, J., 1999. Forensic and environmental detection of explosives. John Wiley & Sons. 304 pgs. ISBN 978-0-471-98371-2
- ^ Haag, W.R., Spanggord, R., Mill, T., Podoll, R.T., Chou, T.W., Tse, D.S., Harper, J.C., 1990. Aquatic environmental fate of nitroguanidine.Environmental Toxicology and Chemistry, 9(11), 1359-1367. doi:10.1002/etc.5620091105
- ^ Boddu, V.M., Abburi, K., Maloney, S.W., Damavarapu, R., 2008. Thermophysical properties of an insensitive munitions compound, 2, 4-dinitroanisole. Journal of Chemical & Engineering Data, 53(5), 1120-1125. doi: 10.1021/je7006764
- ^ Hawari, J., Monteil-Rivera, F., Perreault, N.N., Halasz, A., Paquet, L., Radovic-Hrapovic, Z., Deschamps, S., Thiboutot, S., Ampleman, G., 2015. Environmental fate of 2, 4-dinitroanisole (DNAN) and its reduced products. Chemosphere, 119, 16-23. doi:10.1016/j.chemosphere.2014.05.047
- ^ Spear, R.J., Louey, C.N., Wolfson, M.G., 1989. A preliminary assessment of 3-nitro-1, 2, 4-triazol-5-one (NTO) as an insensitive high explosive (No. MRL-TR-89-18). Materials Research Labs (Australia). Report pdf
- ^ Motzer, W.E., 2001. Perchlorate: problems, detection, and solutions. Environmental Forensics, 2(4), 301-311. doi:10.1006/enfo.2001.0059
- ^ Walsh, M.R., Walsh, M.E., Poulin, I., Taylor, S., Douglas, T.A., 2011. Energetic residues from the detonation of common US ordnance. International Journal of Munitions Constituents and Chemical Propulsion, 10(2). doi: 10.1615/IntJEnergeticMaterialsChemProp.2012004956
- ^ 14.0 14.1 Taylor, S., Hewitt, A., Lever, J., Hayes, C., Perovich, L., Thorne, P., Daghlian, C., 2004. TNT particle size distributions from detonated 155-mm howitzer rounds. Chemosphere, 55(3), 357-367. doi:10.1016/j.chemosphere.2003.11.031
- ^ Taylor, S., Campbell, E., Perovich, L., Lever, J., Pennington, J., 2006. Characteristics of Composition B particles from blow-in-place detonations. Chemosphere, 65(8), 1405-1413. doi: 10.1016/j.chemosphere.2006.03.077
- ^ Dauphin, L., Doyle, C., 2000. Study of ammunition dud and low order detonation rates. US Army Environmental Center. Report SFIM-ACE-ET-CR-200049. Aberdeen Proving Ground, MD.
- ^ Taylor, S., Lever, J., Walsh, M., Walsh, M.E., Bostick, B., Packer, B., 2004. Underground UXO: Are they a significant source of explosives in soil compared to low-and high-order detonations (No. ERDC/CRREL-TR-04-23). Engineer Research and Development Center Hanover NH Cold Regions Research and Engineering Lab. Report pdf
- ^ Taylor, S., K.F. Jones, 2015. The relationship between size and aerial distributions of residues and the detonation yields of rounds. In Munitions Related Contamination, North Atlantic Treaty Organization, Science and Technology Organization Specialists Meeting AVT-244.
- ^ 19.0 19.1 19.2 Taylor S., D.B. Ringelberg, K. Dontsova, C. Daghlian, Walsh, M.E., Walsh, M.R., 2013. Insights into the dissolution and the three-dimensional structure of Insensitive Munitions Formulations. Chemosphere, 93(9), 1782-1788. doi: 10.1016/j.chemosphere.2013.06.011
- ^ 20.0 20.1 20.2 20.3 20.4 Taylor, S., Dontsova, K., Bigl, S., Richardson, C., Lever, J., Pitt, J., Bradley, J.P., Walsh, M., Simunek, J., 2012. Dissolution Rate of Propellant Energetics from Nitrocellulose Matrices (No. ERDC/CRREL-TR-12-9). Engineer Research and Development Center Hanover NH Cold Regions Research And Engineering Lab. ER-1691
- ^ 21.0 21.1 21.2 Taylor, S., Lever, J.H., Fadden, J., Perron, N., Packer, B., 2009. Simulated rainfall-driven dissolution of TNT. Tritonal, Comp B and Octol particles. Chemosphere, 75(8), 1074-1081. doi: 10.1016/j.chemosphere.2009.01.031
- ^ 22.0 22.1 Taylor, S., Lever, J.H., Fadden, J., Perron, N., Packer, B., 2009. Outdoor weathering and dissolution of TNT and Tritonal. Chemosphere, 77(10), 1338-1345. doi:10.1016/j.chemosphere.2009.09.040
- ^ Lever, J.H., Taylor, S., Perovich, L., Bjella, K., Packer, B., 2005. Dissolution of composition B detonation residuals. Environmental Science & Technology, 39(22), 8803-8811. doi:10.1021/es050511r
- ^ 24.0 24.1 24.2 Taylor, S., Park, E., Bullion, K., Dontsova, K., 2015. Dissolution of three insensitive munitions formulations. Chemosphere, 119, 342-348. doi: 10.1016/j.chemosphere.2014.06.050
- ^ Lynch, J.C., Brannon, J.M., Delfino, J.J., 2002. Dissolution rates of three high explosive compounds: TNT, RDX, and HMX. Chemosphere, 47(7), 725-734. doi: 10.1016/s0045-6535(02)00035-8
- ^ Lynch, J.C., Brannon, J.M., Delfino, J.J., 2002. Effects of component interactions on the aqueous solubilities and dissolution rates of the explosive formulations octol, composition B, and LX-14. Journal of Chemical & Engineering Data, 47(3), 542-549. doi: 10.1021/je010294j
- ^ Phelan, J. M., Webb, S. W., Romero, J. V., Barnett, J. L., Griffin, F., Eliassi, M., 2003. Measurement and Modeling of Energetic Material Mass Transfer to Soil Pore Waters. Project CP-1227. Sandia Report 2003-0153. Report pdf
- ^ Hewitt, A.D., Bigl, S.R., 2005. Elution of energetic compounds from propellant and composition B residues (No. ERDC/CRREL-TR-05-13). Engineering Research and Development Center Hanover NH Cold Regions Research and Engineering Lab. Report pdf
- ^ Taylor, S., Dontsova, K., Walsh, M.E., Walsh, M.R., 2015. Outdoor dissolution of detonation residues of three insensitive munitions (IM) formulations. Chemosphere, 134, 250-256. doi:10.1016/j.chemosphere.2015.04.041
- ^ 30.0 30.1 Walsh, M.R., Walsh, M.E., Ramsey, C.A., Brochu, S., Thiboutot, S., Ampleman, G., 2013. Perchlorate contamination from the detonation of insensitive high-explosive rounds. Journal of Hazardous Materials, 262, 228-233. doi: 10.1016/j.jhazmat.2013.08.045
- ^ 31.0 31.1 Walsh, M.R., Walsh, M.E., Ramsey, C.A., Thiboutot, S., Ampleman, G., Dowden, J., 2015. Energetic Residues from the Detonation of IMX101 and IMX-104 Munitions. ERDC/CRREL TR-15-3.
- ^ Richard, T., Weidhaas, J., 2014. Dissolution, sorption, and phytoremediation of IMX-101 explosive formulation constituents: 2, 4-dinitroanisole (DNAN), 3-nitro-1, 2, 4-triazol-5-one (NTO), and nitroguanidine. Journal of Hazardous Materials, 280, 561-569. doi: 10.1016/j.jhazmat.2014.08.042