Difference between revisions of "Main Page"
Debra Tabron (talk | contribs) (Tag: Visual edit) |
|||
| Line 67: | Line 67: | ||
| style="vertical-align:top;" | | | style="vertical-align:top;" | | ||
<u>'''[[Transport & Attenuation Processes | Attenuation & Transport Processes]]'''</u> | <u>'''[[Transport & Attenuation Processes | Attenuation & Transport Processes]]'''</u> | ||
| + | |||
*[[Advection and Groundwater Flow]] | *[[Advection and Groundwater Flow]] | ||
*[[Biodegradation - 1,4-Dioxane]] | *[[Biodegradation - 1,4-Dioxane]] | ||
| Line 82: | Line 83: | ||
<u>'''[[Characterization, Assessment & Monitoring]]'''</u> | <u>'''[[Characterization, Assessment & Monitoring]]'''</u> | ||
| + | |||
*[[Characterization Methods – Hydraulic Conductivity]] | *[[Characterization Methods – Hydraulic Conductivity]] | ||
*[[Compound Specific Isotope Analysis (CSIA)|Compound Specific Isotope Analysis (CSIA)]] | *[[Compound Specific Isotope Analysis (CSIA)|Compound Specific Isotope Analysis (CSIA)]] | ||
| Line 90: | Line 92: | ||
**[[Geophysical Methods - Case Studies | Case Studies]] | **[[Geophysical Methods - Case Studies | Case Studies]] | ||
*[[Groundwater Sampling - No-Purge/Passive]] | *[[Groundwater Sampling - No-Purge/Passive]] | ||
| + | *[[LNAPL Conceptual Site Models]] | ||
*[[Long-Term Monitoring (LTM)|Long-Term Monitoring (LTM)]] | *[[Long-Term Monitoring (LTM)|Long-Term Monitoring (LTM)]] | ||
**[[Long-Term Monitoring (LTM) - Data Analysis | LTM Data Analysis]] | **[[Long-Term Monitoring (LTM) - Data Analysis | LTM Data Analysis]] | ||
| Line 97: | Line 100: | ||
**[[Quantitative Polymerase Chain Reaction (qPCR)]] | **[[Quantitative Polymerase Chain Reaction (qPCR)]] | ||
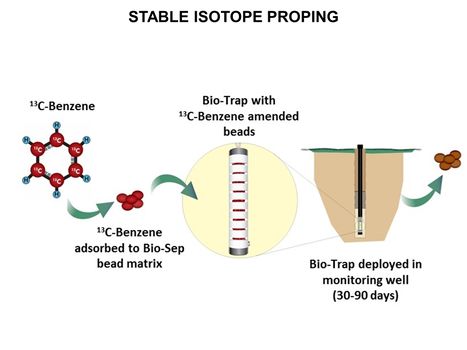
**[[Stable Isotope Probing (SIP)]] | **[[Stable Isotope Probing (SIP)]] | ||
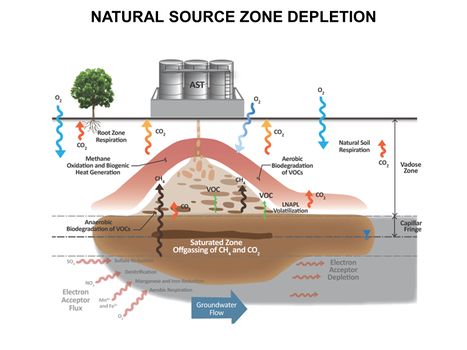
| − | *[[Natural Attenuation in Source Zone and Groundwater Plume - Bemidji Crude Oil Spill | Natural Attenuation in Source Zone and Groundwater Plume -<br/> Bemidji Crude Oil Spill]] | + | *[[Natural Attenuation in Source Zone and Groundwater Plume - Bemidji Crude Oil Spill | Natural Attenuation in Source Zone and Groundwater Plume -<br /> Bemidji Crude Oil Spill]] |
<u>'''[[Climate Change]]'''</u> | <u>'''[[Climate Change]]'''</u> | ||
| + | |||
*[[Climate Change Primer]] | *[[Climate Change Primer]] | ||
| style="width:33%; vertical-align:top; " | | | style="width:33%; vertical-align:top; " | | ||
<u>'''[[Coastal and Estuarine Ecology]]'''</u> | <u>'''[[Coastal and Estuarine Ecology]]'''</u> | ||
| + | |||
*[[Phytoplankton (Algae) Blooms]] | *[[Phytoplankton (Algae) Blooms]] | ||
<u>'''[[Contaminated Sediments - Introduction | Contaminated Sediments]]'''</u> | <u>'''[[Contaminated Sediments - Introduction | Contaminated Sediments]]'''</u> | ||
| + | |||
*[[In Situ Treatment of Contaminated Sediments with Activated Carbon]] | *[[In Situ Treatment of Contaminated Sediments with Activated Carbon]] | ||
<u>'''[[Munitions Constituents]]'''</u> | <u>'''[[Munitions Constituents]]'''</u> | ||
| + | |||
*[[Munitions Constituents - Alkaline Degradation| Alkaline Degradation]] | *[[Munitions Constituents - Alkaline Degradation| Alkaline Degradation]] | ||
*[[Munitions Constituents - Composting| Composting]] | *[[Munitions Constituents - Composting| Composting]] | ||
| Line 122: | Line 129: | ||
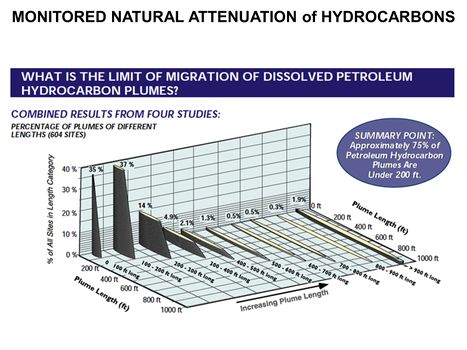
<u>'''[[Monitored Natural Attenuation (MNA)]]'''</u> | <u>'''[[Monitored Natural Attenuation (MNA)]]'''</u> | ||
| + | |||
*[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents| MNA of Chlorinated Solvents]] | *[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents| MNA of Chlorinated Solvents]] | ||
*[[Monitored Natural Attenuation (MNA) of Metal and Metalloids| MNA of Metals and Metalloids]] | *[[Monitored Natural Attenuation (MNA) of Metal and Metalloids| MNA of Metals and Metalloids]] | ||
| Line 129: | Line 137: | ||
<u>'''[[Regulatory Issues and Site Management]]'''</u> | <u>'''[[Regulatory Issues and Site Management]]'''</u> | ||
| + | |||
*[[Alternative Endpoints]] | *[[Alternative Endpoints]] | ||
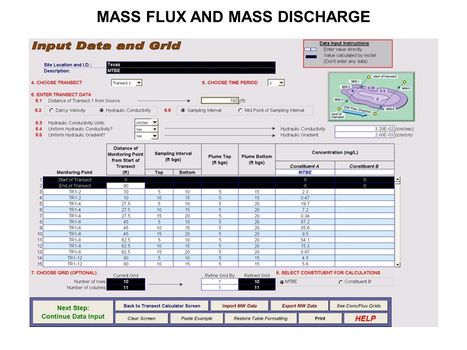
*[[Mass Flux and Mass Discharge]] | *[[Mass Flux and Mass Discharge]] | ||
| Line 168: | Line 177: | ||
<u>'''[[Soil & Groundwater Contaminants]]'''</u> | <u>'''[[Soil & Groundwater Contaminants]]'''</u> | ||
| + | |||
*[[1,4-Dioxane]] | *[[1,4-Dioxane]] | ||
*[[Chlorinated Solvents]] | *[[Chlorinated Solvents]] | ||
Revision as of 13:18, 4 September 2020
Peer Reviewed. Accessible. Written By Experts |
Your Environmental Information Gateway |
| The goal of ENVIRO.wiki is to make scientific and engineering research results more accessible to environmental professionals, facilitating the permitting, design and implementation of environmental projects. Articles are written and edited by invited experts (see Contributors) to summarize current knowledge for the target audience on an array of topics, with cross-linked references to reports and technical literature. | See Table of Contents |
Featured article: In Situ Treatment of Contaminated Sediments with Activated CarbonThe ultraviolet (UV)/sulfite based reductive defluorination process has emerged as an effective and practical option for generating hydrated electrons (eaq- ) which can destroy per- and polyfluoroalkyl substances (PFAS) in water. A UV/sulfite treatment system has been demonstrated in two field demonstrations in which it achieved near-complete defluorination and greater than 99% destruction of 40 PFAS analytes measured by EPA method 1633. Hydrated electrons can be produced by photoirradiation of solutes, including sulfite, iodide, dithionite, and ferrocyanide, and have been reported in literature to effectively decompose per- and polyfluoroalkyl substances (PFAS) in water. The hydrated electron is one of the most reactive reducing species, with a standard reduction potential of about −2.9 volts. Though short-lived, hydrated electrons react rapidly with many species having more positive reduction potentials. Under the Environmental Security Technology Certification Program (ESTCP) Project ER21-5152, a field demonstration was conducted at a Navy site on the east coast, and results showed that the technology was highly effective in destroying various PFAS in a liquid concentrate produced from an in situ foam fractionation groundwater treatment system. Another field demonstration was completed at an Air Force base in California, where a treatment train was used to treat PFAS in groundwater. PFAS analytical data and fluoride results demonstrated near-complete destruction of various PFAS. In addition, this demonstration showed: a) high PFAS destruction ratio was achieved in the foam fractionate, even in very high concentration (up to 1,700 mg/L of booster), and b) the effluent was sent back to the influent of the system for further concentration and treatment, resulting in a closed-loop treatment system and no waste discharge.
(Full article...) |
Enviro Wiki Highlights |